Folic acid fortification: should you be concerned?
Folic acid fortification: should we be concerned?
Since the late 1990s, many countries including Canada and the United States have required the addition of folic acid to refined grain products such as white flour. The goal of this policy was clear: increase folate intake in the population and reduce the risk of neural tube defects (NTDs), serious birth defects affecting the brain and spine. The policy has been widely considered a public health success because rates of NTDs declined after fortification was introduced.
However, as our understanding of folate metabolism improves, scientists are asking a new question: could widespread exposure to synthetic folic acid have unintended consequences?
Folic acid is not the same as natural folate
Folate is a vitamin B9 naturally found in foods such as leafy greens, legumes, and some fruits. In the body, the main active form circulating in the blood is 5-methyltetrahydrofolate (5-MTHF).
Folic acid, by contrast, is a synthetic form of vitamin B9 used in supplements and food fortification because it is inexpensive and stable during food processing. Before the body can use it, folic acid must be converted through several steps into active folate forms.
One of these steps involves an enzyme called dihydrofolate reductase (DHFR). In humans, this enzyme works relatively slowly. When intake of folic acid exceeds the body’s capacity to process it, some of the vitamin can remain in the bloodstream in its original form. This circulating form is called unmetabolized folic acid (UMFA).
Why researchers are paying attention to UMFA
In countries with mandatory fortification, studies have found that UMFA can be detected in the blood of many individuals, particularly when people consume both fortified foods and supplements. The presence of UMFA does not necessarily mean harm, but it indicates that the body’s metabolic capacity has been exceeded.
Researchers are investigating whether long-term exposure to UMFA could have biological effects. One concern is that folic acid binds strongly to certain folate receptors and transport systems, which are responsible for moving folate into cells. Because folic acid can have a higher affinity for some receptors than natural folate forms, scientists have proposed that it could potentially compete with active folates such as 5-MTHF in certain tissues.
This possibility has been discussed particularly in relation to the transport of folate into the brain, which occurs at the blood–cerebrospinal fluid barrier through specialized folate receptors. In rare conditions such as cerebral folate deficiency, researchers have observed that high levels of folic acid may interfere with the transport of active folate into the central nervous system. These findings come mainly from clinical observations and mechanistic studies and do not necessarily reflect what occurs in the general population, but they illustrate that the form of folate can matter biologically.
MTHFR mutations: more at risk of UMFA
While the MTHFR gene polymorphism, affecting up to 40% of the population, is often spotlighted for impairing the conversion of folic acid to its active form, 5-methyltetrahydrofolate (5-MTHF or methylfolate), it’s crucial to recognize that this metabolic bottleneck isn’t isolated to genetic variants alone. In fact, the enzyme dihydrofolate reductase (DHFR), which initiates the reduction of synthetic folic acid at the very beginning of the folate metabolic pathway, operates at remarkably low efficiency in all humans—regardless of MTHFR status.
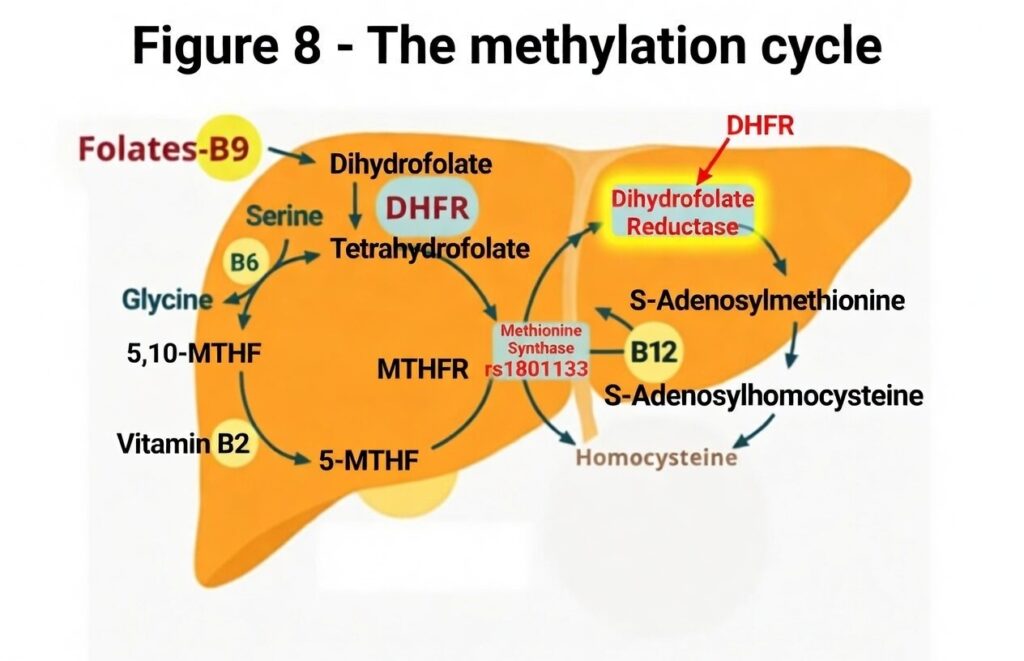
A seminal 1987 study by Whitehead, Kamen, and Beaulieu compared DHFR levels across species and found human liver activity to be strikingly diminished compared to rodents, birds, or even monkeys, processing folic acid up to 1,300 times slower than natural folate substrates. (1)This universal human limitation means that even modest intakes from fortified foods (like grains and cereals, adding 100-200 μg daily) or supplements can overwhelm our capacity, leading to the accumulation of unmetabolized folic acid (UMFA) in the bloodstream.
Effects of unmetabolized folic acid
UMFA doesn’t just linger harmlessly; it competes with methylfolate for cellular uptake via shared transporters like SLC19A1 and receptors such as folate receptor alpha (FRα), which preferentially binds UMFA. This interference disrupts one-carbon metabolism, essential for DNA synthesis, repair, and methylation—processes directly implicated in cancer risk. For instance, elevated UMFA has been linked to reduced natural killer (NK) cell cytotoxicity, impairing the body’s ability to eliminate precancerous cells(2,3). Studies, including a meta-analysis of six trials, associate high folic acid supplementation with a 24% increased prostate cancer risk (4). others report 21% higher overall cancer incidence and 38% elevated mortality in certain cohorts.
Thus, lacking an MTHFR variant offers no false sense of security. Everyone’s sluggish DHFR renders us vulnerable to UMFA’s potential oncogenic effects, especially in a fortified food landscape where additional supplements can exacerbate exposure. To mitigate cancer risks, individuals should prioritize natural folate sources (e.g., leafy greens) or methylfolate supplements, which bypass these inefficiencies. Consulting healthcare providers for personalized folate status assessments is a good idea. In fact, awareness of this broader metabolic constraint underscores that folate management is a universal concern in cancer prevention, not merely a genetic one.
Choose your supplements well
Should you be concerned? If you’re eating a typical Western diet, you’re already getting folic acid without realizing it. Layering on supplements for “extra insurance” could tip you into UMFA territory, fostering silent health issues that manifest years later, like cerebral folate deficiency syndrome. Vulnerable groups are pregnant people or breastfeeding mothers who take prenatal supplement often containing 1000mcg. (5,6) The elderly, or those with genetic variants also face amplified dangers. Alternatives exist. Whether you have a MTHFR variant or not, if you think a supplement is needed, opt for methylfolate supplements and avoid UMFA formation entirely. Emerging studies show they’re equally effective at raising folate levels and may offer superior benefits for those with MTHFR issues.
A question of balance
It is important to remember that folic acid fortification was introduced to address a real public health problem. Before fortification, many women did not consume enough folate to protect against neural tube defects during early pregnancy. Increasing folate intake at the population level contributed to a measurable decline in these conditions.
At the same time, nutrition science continues to evolve. Today, people may receive folic acid from several sources simultaneously: fortified flour products, breakfast cereals, supplements, and prenatal vitamins. This combination can lead to higher total exposure than originally anticipated when fortification policies were designed.
Some researchers therefore argue that it may be time to re-evaluate how folate is delivered to the population. 5-MTHF (methylfolate) the biologically active form of folate that does not produce unmetabolized folic acid in the bloodstream is being studied.
What does this mean for you?
For most people, the best way to support healthy folate status remains simple. Eat a balanced diet rich in natural folate sources, including leafy greens, legumes, and vegetables. These foods provide folate in forms that the body can readily use, along with many other beneficial nutrients, without the risk of accumulating in your blood.
At the population level, the debate about folic acid fortification reflects a broader challenge in nutrition policy: how to design interventions that protect vulnerable groups without creating unintended exposures for others. As research on folate metabolism continues, public health authorities may eventually revisit how best to achieve that balance.
Want to read more about folic acid and the case for neural tube defects? Read this article
References
(1) Whitehead VM, Kamen BA, Beaulieu D. Levels of dihydrofolate reductase in livers of birds, animals, primates, and man. Cancer Drug Delivery. 1987;4(3):185-189. doi:10.1089/cdd.1987.4.185. https://pubmed.ncbi.nlm.nih.gov/3450381/
(2) Troen AM, Mitchell B, Sorensen B, et al. Unmetabolized folic acid in plasma is associated with reduced natural killer cell cytotoxicity among postmenopausal women. The Journal of Nutrition. 2006;136(1):189-194. doi:10.1093/jn/136.1.189. https://pubmed.ncbi.nlm.nih.gov/16365081/
(3) Paniz C. et al. (2017) A Daily Dose of 5 mg Folic Acid for 90 Days Is Associated with Increased Serum Unmetabolized Folic Acid and Reduced Natural Killer Cell Cytotoxicity in Healthy Brazilian Adults. Journal of Nutrition. 2017;147(9):1677-1685. https://pubmed.ncbi.nlm.nih.gov/28724658/
(4) Wien TN, Pike E, Wisløff T, Staff A, Smeland S, Klemp M. Cancer risk with folic acid supplements: a systematic review and meta-analysis. BMJ Open. 2012;2(1):e000653. doi:10.1136/bmjopen-2011-000653. https://pubmed.ncbi.nlm.nih.gov/22240654/
(5) Valera-Gran D, García de la Hera M, Navarrete-Muñoz EM, et al. Effect of maternal high dosages of folic acid supplements on neurocognitive development in children at 4–5 y of age: the prospective birth cohort Infancia y Medio Ambiente (INMA) study. The American Journal of Clinical Nutrition. 2017;106(3):878-887. doi:10.3945/ajcn.117.152769. https://pubmed.ncbi.nlm.nih.gov/28724645/
(6) Cochrane KM, O’Connor DL, da Silva Pereira T, et al. Human milk unmetabolized folic acid is increased following supplementation with synthetic folic acid as compared to (6S)-5-methyltetrahydrofolic acid. The American Journal of Clinical Nutrition. 2023;118(3):576-585. https://pubmed.ncbi.nlm.nih.gov/37438496/